Honors Organic Lab | Spring Semester 2014
Faculty Advisors Dr. Allan Pinhas | Dr. Deborah Lieberman
Above are some of the things that I used frequently in lab. The round flask and the small, clear vial with yellow liquid contains some of the compounds that were produced, and the large vial in the back containing white powder contains a special starting material. The graduated cylinder contains our typical solvent, diethyl ether, and the red metal flask is a high speed ball mill vial.
Hello reader! I have put this page together in order to outline my experience in the University of Cincinnati Honors Organic II Lab. I had the privilege of completing this experience with a small group of other students, and I have provided a detailed overview below. Also, I have attached the final presentation that my research group gave at the end of the semester as an artifact from this experience. I hope you enjoy reading about the lab just as much as I enjoyed completing it! Chemistry is my favorite subject, and in finding an experience centered upon it, I consider myself fortunate.
Scientific Background
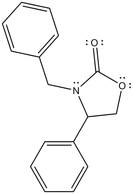
In my lab, the main focus was upon a compound (or rather, a group of compounds) referred to as oxazolidinone. It looks something like this:
In my lab, the main focus was upon a compound (or rather, a group of compounds) referred to as oxazolidinone. It looks something like this:
Before I get explain why oxazolidinone matters, I should first give you some background knowledge. Bear with me. Place your right and left hands next to each other, palms up. Notice that your left hand is a reflected version of your right. Now, palms still up, try to stack your hands on top of each other. It is not possible to align them perfectly. Your hands, in other words, are non-superimposable. More specifically, they are non-superimposable mirror images. When viewed with respect to a mirror plane between the two, they are identical, but you can never perfectly stack them on top of one another when they are facing the same way, i.e. palms up. Your hands, in other words, while they appear the same, are inherently different.
The same phenomenon is seen in organic chemistry. Two molecules may be non-superimposable mirror images of each other, just like your hands. In the world of organic chemistry, we call those two molecules chiral. Chirality is extremely important in organic chemistry, and is even more so with regards to compounds that relate to living things, like medications. Usually, one version (one hand) of a medicine has beneficial effects, while the other version (the other hand) may be completely useless or even harmful. In 2D the molecules are identical, but they are actually inherently different. As a result, organic chemists devote a considerable amount of time to tailoring the way in which they create molecules in order to create the beneficial version of the compound.
With that explained, I can outline the importance of oxazolidinone. Two of its most important uses, as a chiral auxiliary and as a protecting group, help chemists to control the outcome of reactions. As a chiral auxiliary, oxazolidinone helps to control the chirality of the final product or intermediate. In other words, it helps the reaction prefer one version of a compound over another. As a protecting group, a chemist can protect a certain part of a molecule in order to get a reaction to happen at a different place on the molecule. In essence, by correctly using oxazolidinone, scientists may be able to come closer to producing the specific molecule that they believe to be beneficial. In the realm of everyday living, oxazolidinone is also an antibacterial pharmaceutical agent. Bacteria, as of yet, have not built a resistance to this class of compounds, and as this is becoming an ever-increasing problem, oxazolidinone could prove useful in the future as an antibacterial agent.
The same phenomenon is seen in organic chemistry. Two molecules may be non-superimposable mirror images of each other, just like your hands. In the world of organic chemistry, we call those two molecules chiral. Chirality is extremely important in organic chemistry, and is even more so with regards to compounds that relate to living things, like medications. Usually, one version (one hand) of a medicine has beneficial effects, while the other version (the other hand) may be completely useless or even harmful. In 2D the molecules are identical, but they are actually inherently different. As a result, organic chemists devote a considerable amount of time to tailoring the way in which they create molecules in order to create the beneficial version of the compound.
With that explained, I can outline the importance of oxazolidinone. Two of its most important uses, as a chiral auxiliary and as a protecting group, help chemists to control the outcome of reactions. As a chiral auxiliary, oxazolidinone helps to control the chirality of the final product or intermediate. In other words, it helps the reaction prefer one version of a compound over another. As a protecting group, a chemist can protect a certain part of a molecule in order to get a reaction to happen at a different place on the molecule. In essence, by correctly using oxazolidinone, scientists may be able to come closer to producing the specific molecule that they believe to be beneficial. In the realm of everyday living, oxazolidinone is also an antibacterial pharmaceutical agent. Bacteria, as of yet, have not built a resistance to this class of compounds, and as this is becoming an ever-increasing problem, oxazolidinone could prove useful in the future as an antibacterial agent.
Honors Lab Details
During this experience, the research group as a whole focused its attention on a few specific properties of oxazolidinone. We investigated different methods of synthesizing the compound, and examined methods of creating the various intermediate compounds seen throughout the synthesis. We also inquired as to how putting other molecules on the base molecule would affect the overall process. We were focused mainly on identifying and clarifying the general properties of the compound, and in perfecting its synthesis. We did not investigate any of the other properties that I have mentioned previously. However, the information gained this year, in years prior, and in years to come will hopefully clarify the properties of this class of compounds, and will therefore prove useful to any chemist that wishes to pursue the subject in greater depth.
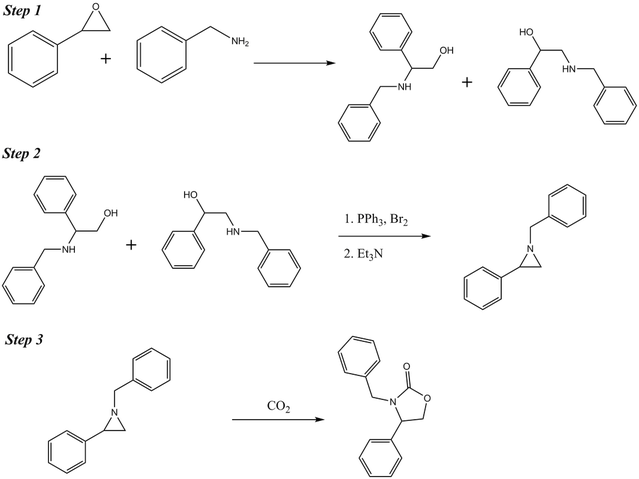
In case you were curious, below is the overall synthesis of oxazolidinone. Unless you’ve taken organic chemistry, this will probably not be helpful at all, but I wanted to throw it in here.
During this experience, the research group as a whole focused its attention on a few specific properties of oxazolidinone. We investigated different methods of synthesizing the compound, and examined methods of creating the various intermediate compounds seen throughout the synthesis. We also inquired as to how putting other molecules on the base molecule would affect the overall process. We were focused mainly on identifying and clarifying the general properties of the compound, and in perfecting its synthesis. We did not investigate any of the other properties that I have mentioned previously. However, the information gained this year, in years prior, and in years to come will hopefully clarify the properties of this class of compounds, and will therefore prove useful to any chemist that wishes to pursue the subject in greater depth.
In case you were curious, below is the overall synthesis of oxazolidinone. Unless you’ve taken organic chemistry, this will probably not be helpful at all, but I wanted to throw it in here.
Throughout the lab, I worked in a team with Cory Wallace and Evan Onusko. Together, we investigated the chirality (there’s that word again - if you are confused, see prior section) of the final step in the synthesis. Above, this is marked as step three. The problem was defined as such: we knew the chirality – the exact version – of the molecule that was produced in steps one and two, but we were missing that piece of information for the final step.
Our first thought was to create a crystal of the compound. In organic chemistry, if you can create a solid crystal that is in reasonably good condition, a crystallographer can tell you the shape of the molecule in 3D space. From this information, we would be able to determine the chirality of the product. We looked into a variety of other methods, but the problem kept surfacing that all of the techniques would tell us how much of each version were created in the reaction, but would not tell us what version was which. I’ll mention these methods a little later. Eventually, on the third last week of lab, we were finally able to get a crystal of our compound, and we sent it to the crystallography lab. However, we obtained the crystal mostly through luck, and we spend the last portion of lab trying to discover a method for explicitly creating a crystal. The problem, we discovered, is that the initial product is impure, and it therefore takes the form of a yellow oil. To purify the oxazolidinone and to produce a crystal, a process called recrystallization is required. However, the details can be quite finicky. Nevertheless, on the last day of lab we had some sort of success. While our professor, Dr. Lieberman, was running tests on one of the crystals, she noticed that the crystal behaved differently with 2-propanol (isopropyl alcohol) than it did with our typical solvent, diethyl ether. We investigated this difference, and were able to create a crude procedure for producing a crystal of oxazolidinone. This method must be verified and perfected next year, but if we are correct, it will allow the final product to be purified in a way not previously possible. Finally, with regards to the crystal that we sent to the lab, the results came back positively after about a month. The crystal that we created was, indeed, oxazolidinone, and that means that our recrystallization method is likely sound. This, above all other things, was one of the most rewarding aspects of the lab, and my group and I are pleased that the crystal was indeed what we had hypothesized.
Our first thought was to create a crystal of the compound. In organic chemistry, if you can create a solid crystal that is in reasonably good condition, a crystallographer can tell you the shape of the molecule in 3D space. From this information, we would be able to determine the chirality of the product. We looked into a variety of other methods, but the problem kept surfacing that all of the techniques would tell us how much of each version were created in the reaction, but would not tell us what version was which. I’ll mention these methods a little later. Eventually, on the third last week of lab, we were finally able to get a crystal of our compound, and we sent it to the crystallography lab. However, we obtained the crystal mostly through luck, and we spend the last portion of lab trying to discover a method for explicitly creating a crystal. The problem, we discovered, is that the initial product is impure, and it therefore takes the form of a yellow oil. To purify the oxazolidinone and to produce a crystal, a process called recrystallization is required. However, the details can be quite finicky. Nevertheless, on the last day of lab we had some sort of success. While our professor, Dr. Lieberman, was running tests on one of the crystals, she noticed that the crystal behaved differently with 2-propanol (isopropyl alcohol) than it did with our typical solvent, diethyl ether. We investigated this difference, and were able to create a crude procedure for producing a crystal of oxazolidinone. This method must be verified and perfected next year, but if we are correct, it will allow the final product to be purified in a way not previously possible. Finally, with regards to the crystal that we sent to the lab, the results came back positively after about a month. The crystal that we created was, indeed, oxazolidinone, and that means that our recrystallization method is likely sound. This, above all other things, was one of the most rewarding aspects of the lab, and my group and I are pleased that the crystal was indeed what we had hypothesized.
Reflection
What made this experience special to me was the manner in which it was conducted. In General Organic Lab, all of the experiments performed have already been perfected, and they are selected to model the concepts introduced in class. For example, we learned about something called Grignard reagents in the second semester, and there is a lab that accompanies this chapter. In general, the experiments selected demonstrate the physical phenomena behind the concepts found in the textbook. Honors Lab, on the other hand, requires the application of knowledge in a different way. In general lab, class learnings are applied by explaining what reaction took place, and by using that knowledge to describe what could have gone wrong. In honors lab, knowledge from class was applied in the more practical sense. We needed to get to a certain place, and we had to use our knowledge to get there—we had to decide how to modify a reaction to improve the result. Also, answers to questions were much less straightforward. If I wondered what would happen when A and B were combined, instead of telling me to refer to page 500 in my lab manual, the professor would say, “I don’t know. Why not just do it and see?”
In essence, the difference between general lab and this experience is the degree of freedom that I was allowed in order to answer the question that had been presented. I wasn’t required to follow a set list of instructions because my group and I were the ones writing the instructions. Along the way we researched methods of recrystallization, lanthanide shift reagents, crystallography, and the use of polarimitry to classify the chirality of organic compounds. We also learned how to use the various spectroscopic instruments available to organic chemists, and we improved our ability to analyze the reports that these machines produce. This research was interesting, informative, and above all, it allowed me to see what it is truly like to be a research chemist. We initially did not have all of the tools we needed to answer the question we were presented with, and instead had to go searching for different methods that would allow us to accomplish our goal. Then, we tried new things based upon the information we gathered. This, to me, is what research is all about.
Upon entering college, one of my main goals was to participate in a research project. I wanted to see what goes on in a research lab, and I wanted to feel what it was like to hypothesize, fail, revise, and try again. Because of this goal, I accepted the offer to join honors lab, and in that respect this experience has exceeded my expectations. Besides investigating and applying advanced concepts that I would never have been exposed to in general lab, I learned more about the process of research. I learned what it felt like to stall out after the first two ideas did not produce the expected outcome. Also, since my grade was based solely on my presentation at the end of the semester, I did not have to be concerned with completing each experiment in perfect detail. I had freedom to focus on the process instead of worrying about how the quality of the final product (or lack thereof) would affect my grade. All of this helped me develop my process oriented thinking skills. I recognized that, in some applications, the process of researching and trying new things is more important than whether or not I actually answer my question in the end. Answering the question would be informative, but what I began to see as the semester progressed was that we learned just as much from our failures as we did from our successes. We knew what did not work, and that almost always told us something, whether it be about the specific reaction or the compound in general. This experience reinforced in my mind the idea that failing at something does not have to be a bad thing, and that it only depends upon what angle the situation is viewed from. I saw that failure can be just as informative – if not more so – as success. In future experiences and classes, this lesson will remain at the forefront of my consciousness, and will allow me to persevere even when things continue to not go as expected.
Another way that this experience will affect my future endeavors is in how it has influenced my views towards research in general. What I have maintained over the years is that I am eager to take part in research, but that I cannot use research as my sole career path. As a result, I have reaffirmed my major of chemical engineering as opposed to chemistry. With chemical engineering, I will be able to further explore my favorite subject and simultaneously apply this information to an ever-changing and dynamic field.
Another important thing this experience has done is reinforce the information I learned in my organic chemistry classes. It forced me to apply my newfound knowledge and use it to critically evaluate what was happening in each reaction. This level of analysis will ensure that the knowledge I gained on my journey through o-chem will remain with me for years to come (I also plan to continue to review this information in small bits so that it will not fade). This experience also taught me how to keep my own research notebook, a skill which will prove useful in industry, and it taught me to work with a research group (as opposed to a class-oriented group). In this group setting, the scope of our involvement was greater than simply finishing an assignment together. Instead, we were able to bounce ideas off of each other in a more relaxed and problem-solving manner. I saw that, in the research process, a group is more than a collection of people – it is the fertile breeding ground of unique and innovative ideas.
The final benefit I received from this experience will help direct my future actions. While I may not wish to pursue research for my entire career, I did see that I enjoy the process enough to continue. As a result, I have secured a research position with another professor in the chemistry department, and next semester I will be looking into something more near and dear to my heart – green chemistry. This experience has allowed me to branch out and become more confident in my organic abilities. It has shown me that I am ready to extend my research and to make it a continual learning process. Honors Organic Lab helped me to define, for myself, what research actually is, where I want to go in the future, and it served as a welcome break from other classes where there was only one right answer. In organic lab, a reaction wasn’t the wrong answer until we tried it and it didn’t work, and this level of freedom allowed my organic knowledge and research abilities to blossom.
I am extremely proud of the method for recrystallization that my group produced. Also, I am pleased that the compound we sent a crystal of was the compound we were searching for, because this means that our group accomplished something previously not possible. It also means that we answered our primary question: what is the chirality of the final step in the reaction? I hope that this will be useful information to those in the future, and I hope to make a contribution this important in my next research experience. Being introduced to the world of green chemistry now will set me up for the future, where I plan to place great emphasis on the environmental aspects of the science that I enjoy so much. It is true that if we destroy our earth while creating new and exciting compounds, nobody will be left to enjoy them. With the knowledge I gained in this experience, I am on my way to making a difference where the science truly excites me. Honors Organic Lab has equipped me with the skills to do research that will, with any luck, help keep our magnificent planet alive and well for years to come.
What made this experience special to me was the manner in which it was conducted. In General Organic Lab, all of the experiments performed have already been perfected, and they are selected to model the concepts introduced in class. For example, we learned about something called Grignard reagents in the second semester, and there is a lab that accompanies this chapter. In general, the experiments selected demonstrate the physical phenomena behind the concepts found in the textbook. Honors Lab, on the other hand, requires the application of knowledge in a different way. In general lab, class learnings are applied by explaining what reaction took place, and by using that knowledge to describe what could have gone wrong. In honors lab, knowledge from class was applied in the more practical sense. We needed to get to a certain place, and we had to use our knowledge to get there—we had to decide how to modify a reaction to improve the result. Also, answers to questions were much less straightforward. If I wondered what would happen when A and B were combined, instead of telling me to refer to page 500 in my lab manual, the professor would say, “I don’t know. Why not just do it and see?”
In essence, the difference between general lab and this experience is the degree of freedom that I was allowed in order to answer the question that had been presented. I wasn’t required to follow a set list of instructions because my group and I were the ones writing the instructions. Along the way we researched methods of recrystallization, lanthanide shift reagents, crystallography, and the use of polarimitry to classify the chirality of organic compounds. We also learned how to use the various spectroscopic instruments available to organic chemists, and we improved our ability to analyze the reports that these machines produce. This research was interesting, informative, and above all, it allowed me to see what it is truly like to be a research chemist. We initially did not have all of the tools we needed to answer the question we were presented with, and instead had to go searching for different methods that would allow us to accomplish our goal. Then, we tried new things based upon the information we gathered. This, to me, is what research is all about.
Upon entering college, one of my main goals was to participate in a research project. I wanted to see what goes on in a research lab, and I wanted to feel what it was like to hypothesize, fail, revise, and try again. Because of this goal, I accepted the offer to join honors lab, and in that respect this experience has exceeded my expectations. Besides investigating and applying advanced concepts that I would never have been exposed to in general lab, I learned more about the process of research. I learned what it felt like to stall out after the first two ideas did not produce the expected outcome. Also, since my grade was based solely on my presentation at the end of the semester, I did not have to be concerned with completing each experiment in perfect detail. I had freedom to focus on the process instead of worrying about how the quality of the final product (or lack thereof) would affect my grade. All of this helped me develop my process oriented thinking skills. I recognized that, in some applications, the process of researching and trying new things is more important than whether or not I actually answer my question in the end. Answering the question would be informative, but what I began to see as the semester progressed was that we learned just as much from our failures as we did from our successes. We knew what did not work, and that almost always told us something, whether it be about the specific reaction or the compound in general. This experience reinforced in my mind the idea that failing at something does not have to be a bad thing, and that it only depends upon what angle the situation is viewed from. I saw that failure can be just as informative – if not more so – as success. In future experiences and classes, this lesson will remain at the forefront of my consciousness, and will allow me to persevere even when things continue to not go as expected.
Another way that this experience will affect my future endeavors is in how it has influenced my views towards research in general. What I have maintained over the years is that I am eager to take part in research, but that I cannot use research as my sole career path. As a result, I have reaffirmed my major of chemical engineering as opposed to chemistry. With chemical engineering, I will be able to further explore my favorite subject and simultaneously apply this information to an ever-changing and dynamic field.
Another important thing this experience has done is reinforce the information I learned in my organic chemistry classes. It forced me to apply my newfound knowledge and use it to critically evaluate what was happening in each reaction. This level of analysis will ensure that the knowledge I gained on my journey through o-chem will remain with me for years to come (I also plan to continue to review this information in small bits so that it will not fade). This experience also taught me how to keep my own research notebook, a skill which will prove useful in industry, and it taught me to work with a research group (as opposed to a class-oriented group). In this group setting, the scope of our involvement was greater than simply finishing an assignment together. Instead, we were able to bounce ideas off of each other in a more relaxed and problem-solving manner. I saw that, in the research process, a group is more than a collection of people – it is the fertile breeding ground of unique and innovative ideas.
The final benefit I received from this experience will help direct my future actions. While I may not wish to pursue research for my entire career, I did see that I enjoy the process enough to continue. As a result, I have secured a research position with another professor in the chemistry department, and next semester I will be looking into something more near and dear to my heart – green chemistry. This experience has allowed me to branch out and become more confident in my organic abilities. It has shown me that I am ready to extend my research and to make it a continual learning process. Honors Organic Lab helped me to define, for myself, what research actually is, where I want to go in the future, and it served as a welcome break from other classes where there was only one right answer. In organic lab, a reaction wasn’t the wrong answer until we tried it and it didn’t work, and this level of freedom allowed my organic knowledge and research abilities to blossom.
I am extremely proud of the method for recrystallization that my group produced. Also, I am pleased that the compound we sent a crystal of was the compound we were searching for, because this means that our group accomplished something previously not possible. It also means that we answered our primary question: what is the chirality of the final step in the reaction? I hope that this will be useful information to those in the future, and I hope to make a contribution this important in my next research experience. Being introduced to the world of green chemistry now will set me up for the future, where I plan to place great emphasis on the environmental aspects of the science that I enjoy so much. It is true that if we destroy our earth while creating new and exciting compounds, nobody will be left to enjoy them. With the knowledge I gained in this experience, I am on my way to making a difference where the science truly excites me. Honors Organic Lab has equipped me with the skills to do research that will, with any luck, help keep our magnificent planet alive and well for years to come.